Dear all,
I have a question regarding the Quantum Yield when used in the Phosphors and Fluorescence Volume Physics section in the non-sequential mode.
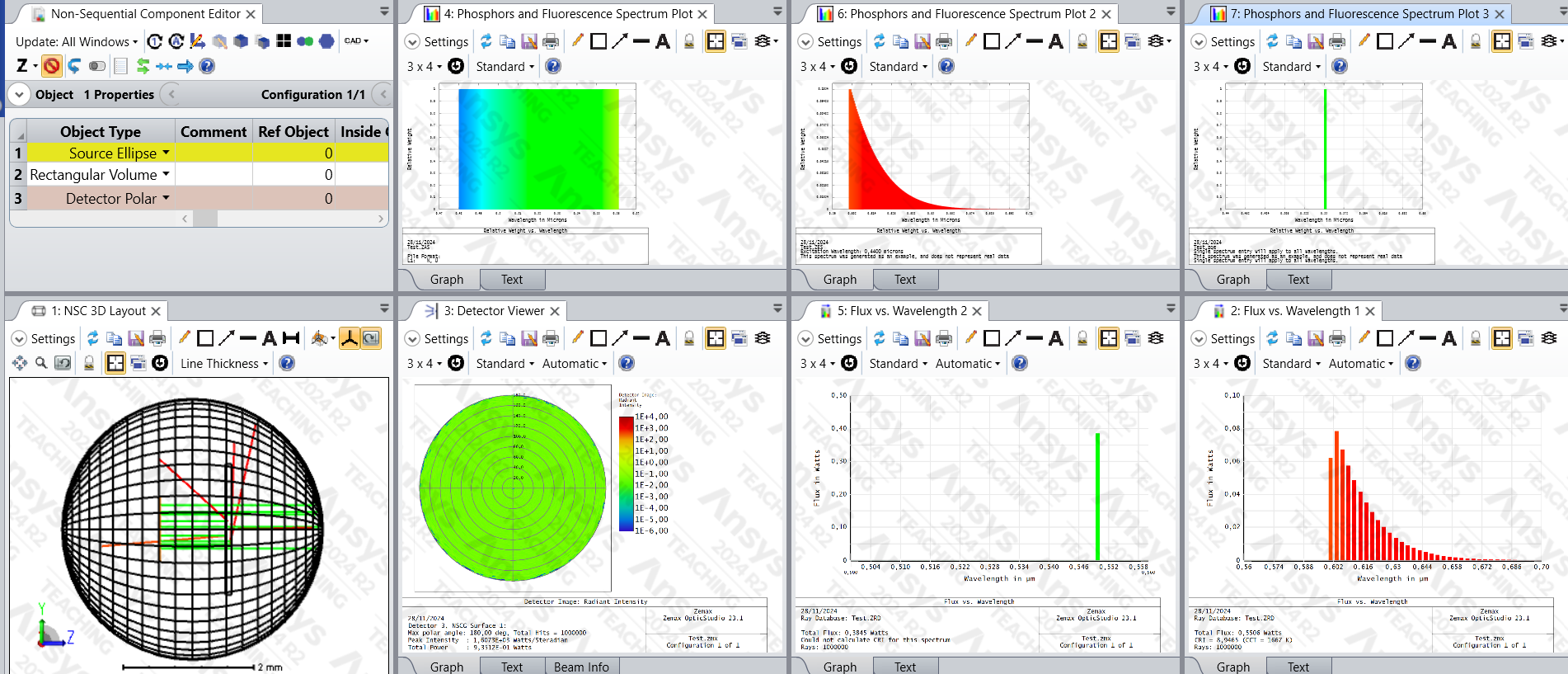
I have a simple setup in Zemax (file in attachment) where I want to replicate a fluorescence quantum yield measurement. It consists of a collimated monochromatic excitation source, a rectangular volume with the volume physics set, and a polar detector capturing all the scattered and fluorescent light. The absorption spectrum is flat at the excitation wavelength (550 nm), the emission spectrum is an arbitrary curve around 600-700 nm, and the quantum yield spectrum is a single value of 1. I have chosen the extinction coefficient such that around 60% of the light is absorbed/re-emitted and the rest is transmitted normally.
With this setup, I would have expected that no light would be lost. However, looking at the total power captured by the polar detector, only 93% of the light remains. Looking at the flux vs wavelength plots shows that we have ~38% of the light at the excitation wavelength and ~55% at the emission wavelengths.
My guess is that the intensity of the fluorescent light is reduced by this ‘Stokes shift’ factor as explained in the documentation:

- Is this indeed the case, or is the reduced power coming from somewhere else?
- If it is the case, what is the reason of including this ratio in this calculation?
- I thought the Internal Quantum Yield Q is always defined as the ratio between the number of re-emitted and absorbed photons (see e.g., https://jascoinc.com/applications/quantum-yield-integrating-sphere/), without any ratio included?
I would be glad if someone could help me understand this issue better.
Thanks,
Best regards,
Indy
